Heat
Heat (q) is the transfer of thermal energy between a system and its surroundings due to a temperature difference. It is the second of the two ways a system can exchange energy with its surroundings.
At the molecular level, thermal energy is the energy of random particle motion, and temperature measures the average kinetic energy of these particles. When objects at different temperatures come into contact, faster-moving particles collide with slower-moving ones, transferring kinetic energy until the averages equalize. This molecular-level energy redistribution is what we observe macroscopically as heat transfer.
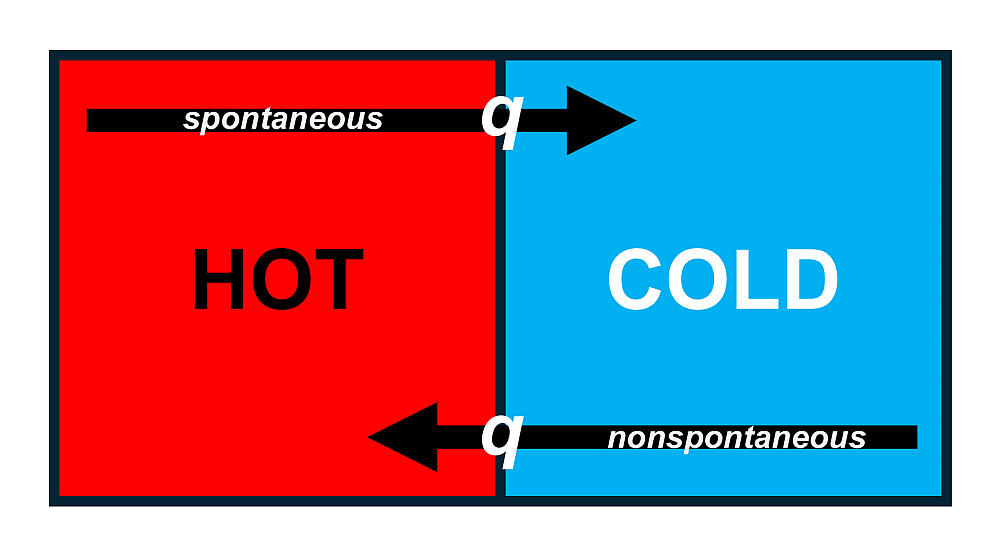
Consider a hot object placed next to a cold object. Our everyday experience tells us that energy will flow from the hot object to the cold one, never the other way around. This is an example of a spontaneous process, one that proceeds in a specific direction without any continuous input of outside energy.
The direction of spontaneous heat transfer is always from a region of higher temperature to a region of lower temperature. A temperature difference is therefore the driving force for this process. This transfer of energy as heat continues until the two objects reach the same temperature, a state known as thermal equilibrium.
The reverse process, where energy is transferred as heat from a cold object to a hot one, is nonspontaneous. It will never happen on its own and can only be forced to occur by doing continuous work on the system, which is the principle behind refrigeration.
Quantifying Heat: The Sign of q
This brings us to a critical point of scientific bookkeeping. When we speak colloquially, we might say that “50 Joules of heat was transferred.” This refers to the magnitude of the energy in transit, which is always a positive number. You cannot transfer a negative amount of energy.
However, in thermodynamics, the variable q is more than just a magnitude. It is an accounting variable that tracks the change in the system’s energy. To do this, q must tell us not only the amount of energy transferred but also its direction.
To understand why, think of your bank account. If you make a transaction of $50, the amount of the transaction is always positive. But to know your final balance, you must know the direction:
- A deposit of $50 is a positive change to your account (+50).
- A withdrawal of $50 is a negative change to your account (−50).
The sign does not mean there is “negative money”; it indicates the direction of the transfer and a change in monetary value of the account.
The variable q works in exactly the same way, and by universal convention, its sign is always determined from the system’s point of view:
- Endothermic Process (q > 0): The system absorbs heat from the surroundings. This is an “energy deposit” into the system, so q is positive.
- This process is spontaneous if the surroundings are hotter than the system.
- Exothermic Process (q < 0): The system releases heat to the surroundings. This is an “energy withdrawal” from the system, so q is negative.
- This process is spontaneous if the system is hotter than the surroundings.
The wording of a problem tells you whether to report a magnitude (always positive) or a signed value (which may be negative). Learning to recognize these cues is essential for clear scientific communication.
Asking for Magnitude (|q|) — answer is always positive:
- “How much heat is released…” (or lost, evolved, produced)
- “How much heat is absorbed…” (or gained)
- “How much heat is transferred…”
- “How much energy is required…” (or needed)
- “What is the amount of energy involved…”
The directional verb tells you which way the energy went; the number itself is always positive.
Asking for the Signed Variable (q) — answer must have the correct sign:
- “What is the value of q…”
- “Determine q…” (or calculate q, find q)
- “What is ΔH for this reaction…”
This sign convention is the foundation that allows us to build a simple energy balance equation, the First Law of Thermodynamics.
Quantifying Heat: Heat Capacity
When energy is transferred as heat to a substance, its temperature increases. However, different substances respond to the same amount of heat in different ways. This inherent property of a substance to resist a change in temperature is quantified by its heat capacity (C) and is temperature dependent.
The specific heat capacity of a substance is not a true constant; it changes with temperature. However, for most solids and liquids over moderate temperature ranges, this variation is small.
For this reason, it is standard practice in general chemistry to use the value of cp measured at 25 °C (298.15 K) and assume it is constant for calculations. Later in this section, we will compare this constant cp approximation with the more rigorous integral calculation and show that our simplified method is highly accurate for typical problems.
Because most chemical reactions in the lab occur at constant atmospheric pressure, we are almost always interested in the heat capacity at constant pressure (Cp). This is an extensive property, meaning it depends on the amount of the substance.
Quick Reference
The Heat Capacity reference tables provide molar (Cp,m) and specific (cp) heat capacities of select elements and compounds at various temperatures.
To have a more useful, intrinsic property, we define two intensive properties derived from Cp:
- The specific heat capacity at constant pressure (cp) is the quantity of heat required to raise the temperature of one gram of a substance by one degree Celsius. Its units are J g−1 °C−1 or J g−1 K−1.
- The molar heat capacity (Cp,m) is the quantity of heat required to raise the temperature of one mole of a substance by one Kelvin at constant pressure. Its units are J mol−1 °C−1 or J mol−1 K−1.
All heat capacity equations assume no phase change occurs during the temperature interval. If the substance melts, boils, or freezes, additional energy terms are needed. These are covered in Enthalpy.
In most laboratory settings, mass is easier to measure than moles, so we will primarily use specific heat capacity (cp). Rearranging its definition provides the central equation for calculating the heat transferred for a substance undergoing a temperature change at constant pressure: \[ q = m c_{\mathrm{p}} \Delta T \]
where
- q is the quantity of heat transferred (in J).
- m is the mass of the substance (in g).
- cp is the specific heat capacity (in J g−1 °C−1 or J g−1 K−1).
- ΔT is the change in temperature (Tfinal − Tinitial) in °C or K.
When working with molar quantities rather than mass, the equivalent relationship uses molar heat capacity:
\[ q = n C_{\mathrm{p,m}} \Delta T \]
where
- n is the amount of substance (in mol).
- Cp,m is the molar heat capacity (in J mol−1 K−1).
Strictly speaking, these equations give qp, the heat transferred at constant pressure. However, since the constant-pressure condition is already indicated by using cp or Cp,m, the subscript on q is typically dropped. Throughout this text (and most others), q in heat capacity contexts means qp.
Because the Celsius and Kelvin scales have the same degree size, a change in temperature (ΔT) has the same numerical value on both scales. A change of 10 °C is identical to a change of 10 K. For this reason, the units for specific heat capacity, J g−1 °C−1 and J g−1 K−1, are directly interchangeable.
Example: Calculate the quantity of heat required to raise the temperature of a 15.0 g block of solid aluminum from 25.0 °C to 35.0 °C. The specific heat capacity of solid aluminum is 0.897 J g−1 °C−1. (Note: This is the accepted reference value for the specific heat capacity at 25 °C (298.15 K). For this problem, we assume the value is constant over the temperature range.)
\[ \begin{align*} q &= m c_{\mathrm{p}} \Delta T \\[1.5ex] &= \left( 15.\bar{0}~\mathrm{g} \right) \left( \dfrac{0.89\bar{7}~\mathrm{J}}{\mathrm{g~^\circ C}} \right) \left( 35.\bar{0}~^\circ\mathrm{C} - 25.\bar{0}~^\circ\mathrm{C} \right) \\[1.5ex] &= \left( 15.\bar{0}~\mathrm{g} \right) \left( \dfrac{0.89\bar{7}~\mathrm{J}}{\mathrm{g~^\circ C}} \right) \left( 10.\bar{0}~^\circ\mathrm{C} \right) \\[1.5ex] &= 13\bar{4}.55~\mathrm{J} \\[1.5ex] &= 135~\mathrm{J} \end{align*} \]
Heat Transfer and Thermal Equilibrium
The equation q=mcpΔT can determine the heat required to change the temperature of a single substance. However, its full utility is revealed when we analyze the transfer of heat between substances.
Consider a hot object placed in contact with a cold object in a perfectly insulated container (a thermally isolated system). Energy will spontaneously transfer as heat from the hot object to the cold one until they reach thermal equilibrium.
Because the system is isolated, the total energy change is zero. This means that the amount of heat lost by the hot object is perfectly balanced by the amount of heat gained by the cold object. This gives us a foundational principle of calorimetry: \[ q_{\mathrm{hot}} + q_{\mathrm{cold}} = 0 \] While correct, a more intuitive way to write this relationship is by rearranging the terms: \[ q_{\mathrm{cold}} = -q_{\mathrm{hot}} \] This simple equation is the key to solving all thermal equilibrium problems. It states: The heat gained by the cold object is equal in magnitude but opposite in sign to the heat released by the hot object.
We can now combine this principle with the heat capacity equation to solve for unknown variables, such as the final temperature of a mixture.
Example: A 55.0 g piece of iron (cp(Fe) = 0.449 J g−1 °C−1) is heated to an initial temperature of 99.8 °C and is then dropped into a beaker containing 225.0 g of water (cp(H2O) = 4.184 J g−1 °C−1) initially at 21.0 °C. Assuming the beaker is a perfect insulator, determine the following:
- The final temperature (in °C) of the system when it reaches thermal equilibrium
- The amount of heat transferred (in J) to reach thermal equilibrium
Note that the provided specific heat capacities are measured at 298.15 K.
Solution
1. Determine the final temperature
$$ \[\begin{align*} \textcolor{royalblue}{q_{\mathrm{water}}} &= -\textcolor{sienna}{q_{\mathrm{iron}}} \\[1.5ex] \textcolor{royalblue}{m_{\mathrm{H_2O}}c_{\mathrm{p, H_2O}}\Delta T_{\mathrm{H_2O}}} &= -\textcolor{sienna}{m_{\mathrm{Fe}}c_{\mathrm{p, Fe}}\Delta T_{\mathrm{Fe}}} \\[1.5ex] \textcolor{royalblue}{m_{\mathrm{H_2O}}c_{\mathrm{p, H_2O}}}(T_{\mathrm{f}} - \textcolor{royalblue}{T_{\mathrm{i, H_2O}}}) &= -\textcolor{sienna}{m_{\mathrm{Fe}}c_{\mathrm{p, Fe}}}(T_{\mathrm{f}} - \textcolor{sienna}{T_{\mathrm{i, Fe}}}) \\[1.5ex] \textcolor{royalblue}{m_{\mathrm{H_2O}} c_{\mathrm{p, H_2O}}}T_{\mathrm{f}} - \textcolor{royalblue}{m_{\mathrm{H_2O}} c_{\mathrm{p, H_2O}}T_{\mathrm{i, H_2O}}} &= -\textcolor{sienna}{m_{\mathrm{Fe}} c_{\mathrm{p, Fe}}}T_{\mathrm{f}} + \textcolor{sienna}{m_{\mathrm{Fe}} c_{\mathrm{p, Fe}}T_{\mathrm{i, Fe}}} \\[1.5ex] \textcolor{royalblue}{m_{\mathrm{H_2O}} c_{\mathrm{p, H_2O}}}T_{\mathrm{f}} + \textcolor{sienna}{m_{\mathrm{Fe}} c_{\mathrm{p, Fe}}}T_{\mathrm{f}} &= \textcolor{sienna}{m_{\mathrm{Fe}} c_{\mathrm{p, Fe}}T_{\mathrm{i, Fe}}} + \textcolor{royalblue}{m_{\mathrm{H_2O}} c_{\mathrm{p, H_2O}} T_{\mathrm{i, H_2O}}} \\[1.5ex] T_{\mathrm{f}}(\textcolor{royalblue}{m_{\mathrm{H_2O}} c_{\mathrm{p, H_2O}}} + \textcolor{sienna}{m_{\mathrm{Fe}} c_{\mathrm{p, Fe}}}) &= \textcolor{sienna}{m_{\mathrm{Fe}} c_{\mathrm{p, Fe}}T_{\mathrm{i, Fe}}} + \textcolor{royalblue}{m_{\mathrm{H_2O}} c_{\mathrm{p, H_2O}} T_{\mathrm{i, H_2O}}} \\[1.5ex] T_{\mathrm{f}} &= \dfrac{\textcolor{sienna}{m_{\mathrm{Fe}} c_{\mathrm{p, Fe}}T_{\mathrm{i, Fe}}} + \textcolor{royalblue}{m_{\mathrm{H_2O}} c_{\mathrm{p, H_2O}}T_{\mathrm{i, H_2O}}}} {\textcolor{royalblue}{m_{\mathrm{H_2O}} c_{\mathrm{p, H_2O}}} + \textcolor{sienna}{m_{\mathrm{Fe}} c_{\mathrm{p, Fe}}}} \\[1.5ex] &= \dfrac{\bigr[ (\textcolor{sienna}{55.\bar{0}~\mathrm{g}})(\textcolor{sienna}{0.44\bar{9}~\mathrm{J~g^{-1}~^\circ C^{-1}}})(\textcolor{sienna}{99.\bar{8}~^\circ\mathrm{C}})\bigr] ~+~ \bigr[ (\textcolor{royalblue}{225.\bar{0}~\mathrm{g}})(\textcolor{royalblue}{4.18\bar{4}~\mathrm{J~g^{-1}~^\circ C^{-1}}})(\textcolor{royalblue}{21.\bar{0}~^\circ\mathrm{C}})\bigr]} {\bigr[ (\textcolor{royalblue}{225.\bar{0}~\mathrm{g}})(\textcolor{royalblue}{4.18\bar{4}~\mathrm{J~g^{-1}~^\circ C^{-1}}})\bigr] ~+~ \bigr[ (\textcolor{sienna}{55.\bar{0}~\mathrm{g}})(\textcolor{sienna}{0.44\bar{9}~\mathrm{J~g^{-1}~^\circ C^{-1}}})\bigr]} \\[1.5ex] &= \dfrac{\textcolor{sienna}{24\bar{6}4.56~\mathrm{J}} + \textcolor{royalblue}{19\bar{7}69.4~\mathrm{J}}} {\textcolor{royalblue}{941.\bar{4}0~\mathrm{J~^\circ C^{-1}}} + \textcolor{sienna}{24.\bar{6}95~\mathrm{J~^\circ C^{-1}}}} \\[1.5ex] &= \dfrac{22\bar{2}33.96~\mathrm{J}}{966.\bar{0}95~\mathrm{J~^\circ C^{-1}}} \\[1.5ex] &= 23.\bar{0}1425~^\circ\mathrm{C} \\[1.5ex] &= 23.0~^\circ\mathrm{C} \end{align*}\] $$
2. Determine the amount of heat transferred
We know that
\[ q_{\mathrm{H_2O}} = -q_{\mathrm{Fe}} \]
We can find qFe directly from the iron’s temperature change:
\[ \begin{align*} q_{\mathrm{Fe}} &= mc_{\mathrm{p}}\Delta T \longrightarrow \\[1.5ex] q_{\mathrm{Fe}} &= mc_{\mathrm{p}}\Delta T \\[1.5ex] &= \left ( 55.\bar{0}~\mathrm{g} \right ) \left (\dfrac{0.44\bar{9}~\mathrm{J}}{\mathrm{g~^{\circ}C}} \right ) \left ( 23.\bar{0}1425~^{\circ}\mathrm{C} - 99.\bar{8}~^{\circ}\mathrm{C}\right ) \\[1.5ex] &= \left ( 55.\bar{0}~\mathrm{g} \right ) \left (\dfrac{0.44\bar{9}~\mathrm{J}}{\mathrm{g~^{\circ}C}} \right ) \left ( -76.\bar{7}85~^{\circ}\mathrm{C}\right ) \\[1.5ex] &= -18\bar{9}6.2~\mathrm{J} \\[1.5ex] &= -19\bar{0}0~\mathrm{J} \\[1.5ex] &= -1.90\times 10^{3}~\mathrm{J} \end{align*} \]
We can verify this result from the water’s perspective:
\[ \begin{align*} q_{\mathrm{H_2O}} &= mc_{\mathrm{p}}\Delta T \\[1.5ex] &= \left ( 225.\bar{0}~\mathrm{g} \right ) \left (\dfrac{4.18\bar{4}~\mathrm{J}}{\mathrm{g~^{\circ}C}} \right ) \left ( 23.\bar{0}1425~^{\circ}\mathrm{C} - 21.\bar{0}~^{\circ}\mathrm{C}\right ) \\[1.5ex] &= \left ( 225.\bar{0}~\mathrm{g} \right ) \left (\dfrac{4.18\bar{4}~\mathrm{J}}{\mathrm{g~^{\circ}C}} \right ) \left ( 2.\bar{0}1425~^{\circ}\mathrm{C}\right ) \\[1.5ex] &= 1\bar{8}96.2~\mathrm{J} \\[1.5ex] &= 1\bar{9}00~\mathrm{J} \\[1.5ex] &= 1.9\times 10^{3}~\mathrm{J} \end{align*} \]
Notice that the energy lost by the iron equals the energy gained by the water. This quantity is the amount of energy that was transferred.
In the example problem above, we used the equation q=mcpΔT and assumed that the specific heat capacity of iron, cp(Fe), was a constant value of 0.449 J g−1 °C−1. From a rigorous technical standpoint, this is a simplification. Specific heat capacity is actually a function of temperature, written as cp(T). So, how much of an error did our simplification introduce?
The Rigorous Calculation (The Integral Approach)
The technically correct way to calculate the heat transferred is to integrate the cp(T) function over the temperature range of the experiment. This is mathematically equivalent to finding the “area under the curve” of a plot of heat capacity versus temperature. \[ q = m \int_{T_1}^{T_2} c_p(T) \,dT \]
The Algebraic Approximation (The Constant cp Approach)
Our simple formula, q=mcpΔT, effectively calculates the heat as the area of a rectangle, where the height of the rectangle is the single value of cp that we assume is constant.
A Visual Comparison
The plot below brings this comparison to life using the data from our example problem for the 55.0 g piece of iron cooling from 99.8 °C to 23.0 °C.
The plot on the left shows the “true” heat calculated by integrating under the actual, slightly curved cp(T) function, giving q = −1954.8 J. The plot on the right shows the heat calculated using our simpler algebraic method, assuming a constant cp value of 0.449 J g−1 °C−1, which gives q = −1896.2 J.
The difference between the two results is only 3.0 %.
As the plot visually demonstrates, the error introduced by assuming the heat capacity is constant is relatively small for this typical problem. The “area of the rectangle” provides a reasonable approximation of the true “area under the curve.” This is why using standard cp values in the equation q = mcpΔT is an accepted practice in general chemistry.
Heat Capacity of an Object
Sometimes, we are interested in the heat capacity of an entire object, which may be composed of multiple materials. In this case, we use the heat capacity (Cp), an extensive property that represents the heat required to raise the temperature of the entire object by 1 °C. Its units are J °C−1 or J K−1. \[ q = C_{\mathrm{p}} \Delta T \] This quantity is most often used to describe the heat capacity of a specific piece of equipment, such as a calorimeter.
In this text, we adhere to IUPAC standards for heat capacity notation:
- Cp (uppercase): Heat capacity at constant pressure (J K−1) - an extensive property
- Example: Cp(calorimeter) = 8.0931 kJ °C−1
- Cp,m (uppercase with m subscript): Molar heat capacity at constant pressure (J mol−1 K−1)
- Example: Cp,m(H2O, l) = 75.35 J mol−1 K−1
- cp (lowercase): Specific heat capacity at constant pressure (J g−1 K−1)
- Example: cp(H2O) = 4.184 J g−1 °C−1
The “p” subscript indicates constant pressure, distinguishing these from constant-volume heat capacities (CV, cV). Since general chemistry almost always operates at constant (atmospheric) pressure, many sources drop the “p” subscript entirely, writing simply C, c, or Cm. You’ll encounter this shorthand on ACS exams and in other textbooks. When in doubt, check the units to identify the quantity.
For solids and liquids, Cp ≈ CV because these phases are nearly incompressible—their volume changes negligibly with temperature, so the work of expansion against external pressure is minimal. This means you can safely use tabulated cp values for condensed phases regardless of whether your process occurs at constant pressure or constant volume.
Summary
Heat and Its Direction
Heat (q) is the transfer of thermal energy due to a temperature difference. Energy spontaneously transfers as heat from hot to cold until thermal equilibrium is reached.
The variable q is a signed quantity from the system’s point of view:
- Endothermic (q > 0): System absorbs heat from surroundings
- Exothermic (q < 0): System releases heat to surroundings
Heat Capacity
Heat capacity quantifies how much heat is required to change a substance’s temperature:
- Heat capacity (Cp): Extensive property for an entire object (J K−1)
- Molar heat capacity (Cp,m): Heat per mole per degree (J mol−1 K−1)
- Specific heat capacity (cp): Heat per gram per degree (J g−1 °C−1)
The central equations for heat transfer at constant pressure:
\[ q = m c_{\mathrm{p}} \Delta T \qquad \text{or} \qquad q = n C_{\mathrm{p,m}} \Delta T \]
Thermal Equilibrium
When two objects at different temperatures are placed in thermal contact within an isolated system:
\[ q_{\mathrm{cold}} = -q_{\mathrm{hot}} \]
The heat gained by the cold object equals the heat lost by the hot object. This principle allows calculation of final equilibrium temperatures.